 |
Home |
Fact Sheet on Secondhand Smoke (cont.) |
| James. Repace, MSc., Physicist
Ichiro Kawachi, PhD, Associate Professor Stanton Glantz, PhD., Professor 9/1/99 |
 |
Home |
Fact Sheet on Secondhand Smoke (cont.) |
| James. Repace, MSc., Physicist
Ichiro Kawachi, PhD, Associate Professor Stanton Glantz, PhD., Professor 9/1/99 |
SHS is a complex mixture of gas and particle-phase chemicals generated during the burning and smoking of tobacco products (CalEPA, 1997). Chemicals present in SHS include irritants and systemic toxicants such as hydrogen cyanide and sulfur dioxide, mutagens and carcinogens such as benzo(a)pyrene, formaldehyde and 4-aminobiphenyl, and reproductive toxicants such as nicotine, cadmium, and carbon monoxide (CalEPA, 1997). Many SHS constituents have been identified as hazardous by state, federal, and international agencies. To date, over 50 compounds in tobacco smoke have been identified as carcinogens and six as developmental or reproductive toxicants by the State of California. Table 2 shows 43 known or suspected carcinogens in tobacco smoke identified by the International Agency for Research on Cancer (IARC, 1987).
| acetaldehyde | dibenzo(a,i)pyrene | N-nitrosdi-n-propylamine |
| acrylonitrile | dibenzo(a,e)pyrene | N-nitrosopyrrolidine |
| arsenic | dibenzo(a,l)pyrene | N-nitrosodi-n-butylamine |
| benz(a)anthracene | dibenzo(a,h)pyrene | ortho-toluidine |
| benzene | formaldehyde | styrene |
| benzo(a)pyrene | hydrazine | urethane |
| benzo(b)fluoranthene | indeno(1,2,3,-cd)pyrene | vinyl chloride |
| benzo(k)fluoranthene | lead | 1,1-dimethylhydrazine |
| cadmium | nickel | 2-nitropropane |
| chromium VI | N-nitrosodiethanolamine | 2-napthylamine |
| DDT | N-nitrosodiethylamine | 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone [NNK] |
| dibenz(a,h)acridine | N'-nitrosodimethylamine | 4-aminobiphenyl |
| dibenz(a,j)acridine | N'nitrosonornicotine | 5-methychrysene |
| dibenz(a,h)anthracene | N-nitrosopiperidine | 7H-dibenzo(c,g)carbazole |
These include aromatic hydrocarbons, di- and polycyclic aromatic hydrocarbons, aldehydes, n-nitroso compounds, polycyclic aza-arenes, other nitrogen compounds, pesticides, halogenated compounds, and heavy metals, which include a variety of human or animal organ-specific carcinogens, e.g., arsenic, cadmium, chromium, nickel, and NNK (lung cancer), benzene (leukemia), formaldehyde and nickel (nasal sinus), 2-naphthylamine and 4-aminobiphenyl (bladder), lead (renal), vinyl chloride (liver).
Exposure assessment of SHS is critical in evaluating the magnitude of individual or group risk, and the effectiveness of strategies to reduce exposure. Exposure to SHS can be assessed through measurement of SHS markers such as respirable particles (RSP) or nicotine in indoor air, SHS biomarkers in saliva, urine, or blood, or through the use of models (Repace et al., 1998; CalEPA, 1997; Jaakkola and Jaakkola, 1997; Repace, 1987; Repace and Lowrey, 1993;). Often individuals are unaware of exposure, particularly outside the home (Cal EPA, 1997) making ascertainment of an adequate control group in epidemiological investigations difficult or impossible (Repace and Lowrey, 1985). Under typical conditions of smoking, building occupancy, and ventilation, indoor smoking produces levels of RSP far in excess of the U.S. federal fine particle standard for particulate matter 2.5 microns in diameter or below, 15 micrograms per cubic meter (mg/m3) [See Appendix A.] This standard is designed to provide increased protection against a wide range of PM-related health effects, including premature mortality and increased hospital admissions and emergency room visits, primarily in the elderly and individuals with cardiopulmonary disease; increased respiratory symptoms and disease, in children and individuals with cardiopulmonary disease such as asthma; decreased lung function, particularly in children and individuals with asthma; and alterations in lung tissue and structure and in respiratory tract defense mechanisms.
To illustrate the effect of this air pollution on restaurant and bar workers, Eisner et al.(1998) studied the association between ETS exposure and respiratory symptoms in a cohort of 53 bartenders before and after California�s prohibition on smoking in all bars and taverns. 74% of the bartenders initially reported respiratory symptoms; of those symptomatic at baseline, 59% no longer had symptoms at follow-up. 77% initially reported sensory irritation symptoms; at follow-up, 78% of these had symptom resolution. After ETS exposure completely ceased, objective measures of pulmonary function showed marked a 5% to 7% improvement after only one month. Eisner et al. (1998) conclude that establishment of smoke-free bars and taverns was associated with improvement of respiratory health.
It is sometimes important to estimate SHS occupational hazards for specific groups of workers, such as flight attendants and casino workers, who have sued the tobacco industry for damages to their health. The risks of passive smoking can be estimated for groups of individuals based upon levels of the nicotine metabolite, cotinine, in blood, saliva, or urine (Repace and Lowrey, 1993; Repace et al., 1998) [Appendix B]. For example, Repace et al. (1998) estimated the risk of fatal lung cancer and heart disease from SHS exposure for office workers as a function of salivary cotinine; as illustrated in Figure 8 below. The horizontal axis shows the cotinine level. In a group of 89 office workers in the State of Rhode Island the median cotinine level was 0.5 ng/ml (Emmons et al., 1994). The vertical axis shows the estimated excess working lifetime exess risk (i.e., probability) of fatal lung cancer or fatal heart disease associated with that level.
U.S. federal occupational and environmental health regulatory decision rules for assessing the harm of environmental agents are shown for comparison. The de minimis risk level (1 x 10-6) corresponds to an excess lifetime risk of fatality of one death per million persons at risk, and is considered "acceptable" from a regulatory standpoint. De manifestis risks (3 x 10-4) are those that are so high that U.S. federal regulatory agencies almost always act to reduce them (Travis et al.(1990). A very unsafe level is the "significant risk," level which is often used as a benchmark by the U.S. Occupational Safety and Health Administration (OSHA, 1994). Risks after control are either reduced to zero, or to below the de minimis risk level.
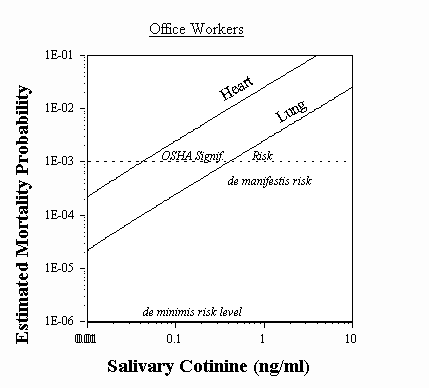
Lifetime risk of mortality for a group of office workers in workplaces with unrestricted smoking estimated as a function of salivary cotinine. For heart disease and lung cancer mortality combined, more than 95% of U.S. nonsmoking office workers exposed under such conditions are estimated to exceed highly unsafe (i.e., significant risk ) occupational regulatory levels. The de manifestis risk level is the obligatory regulatory level and the de minimis levels is the level of maximum acceptable risk (Repace et al., 1998).
Control: Why are ventilation, spatial separation, or air cleaning not valid control measures for SHS, as the tobacco and hospitality industries so often assert? Spatial separation of smokers from nonsmokers within a space does not affect either the smoker density nor the ventilation rate, and so cannot reduce the average SHS concentration. Insofar as ventilation or air cleaning, the U.S. OSHA(1994) has stated: "The carcinogenicity of [SHS] discounts the use of general ventilation as an engineering control for this contaminant." This is illustrated in Figure 9 below (Repace et al., 1998). To achieve de minimis risk would require in excess of one hundred thousand cubic feet per minute per occupant (50,000 litres per second per occupant), which would need tornado-like levels of air flow to achieve. Even if SHS is treated as as simple respirable particulate air pollution, for comparison with the U.S. National Ambient Air Quality Standard (NAAQS) for fine particles (PM2.5), it is clear that ventilation designed essentially to limit carbon dioxide from human metabolism is incapable of controlling massive clouds of tobacco tar particles (See Appendix A).
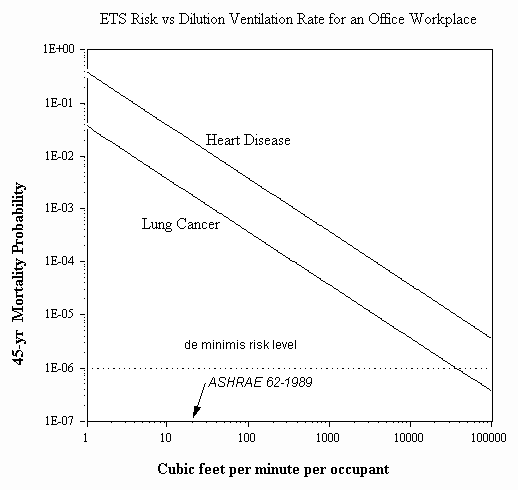
Ventilation cannot control SHS to an acceptable i.e., de minimis, risk level (Repace et al., 1998). Working lifetime risk for office workers exposed to unrestricted smoking (~2 smokers per 100 m2 or 1000 ft2 of office space) versus mechanical ventilation rate. A mechanical ventilation rate of 10 litres per second per occupant is equivalent to 20 cubic feet per minute per occupant (the ventilation rate recommended for offices and restaurants by the cognizant engineering authority in North America, The American Society for Heating, Refrigerating, and Ventilating Engineers (ASHRAE, 1989).
[ Abstract ]
[ Introduction ]
[ Secondhand Smoke and Cardiovascular Disease ]
Secondhand Smoke Exposure
[ Smoke-Free Restaurant Laws ]
[ SHS Lessons from California ]
[ Conclusions ]
[ Technical Appendices ]